Phase 0 approaches also have the potential to provide data that is not readily available with traditional approaches. 
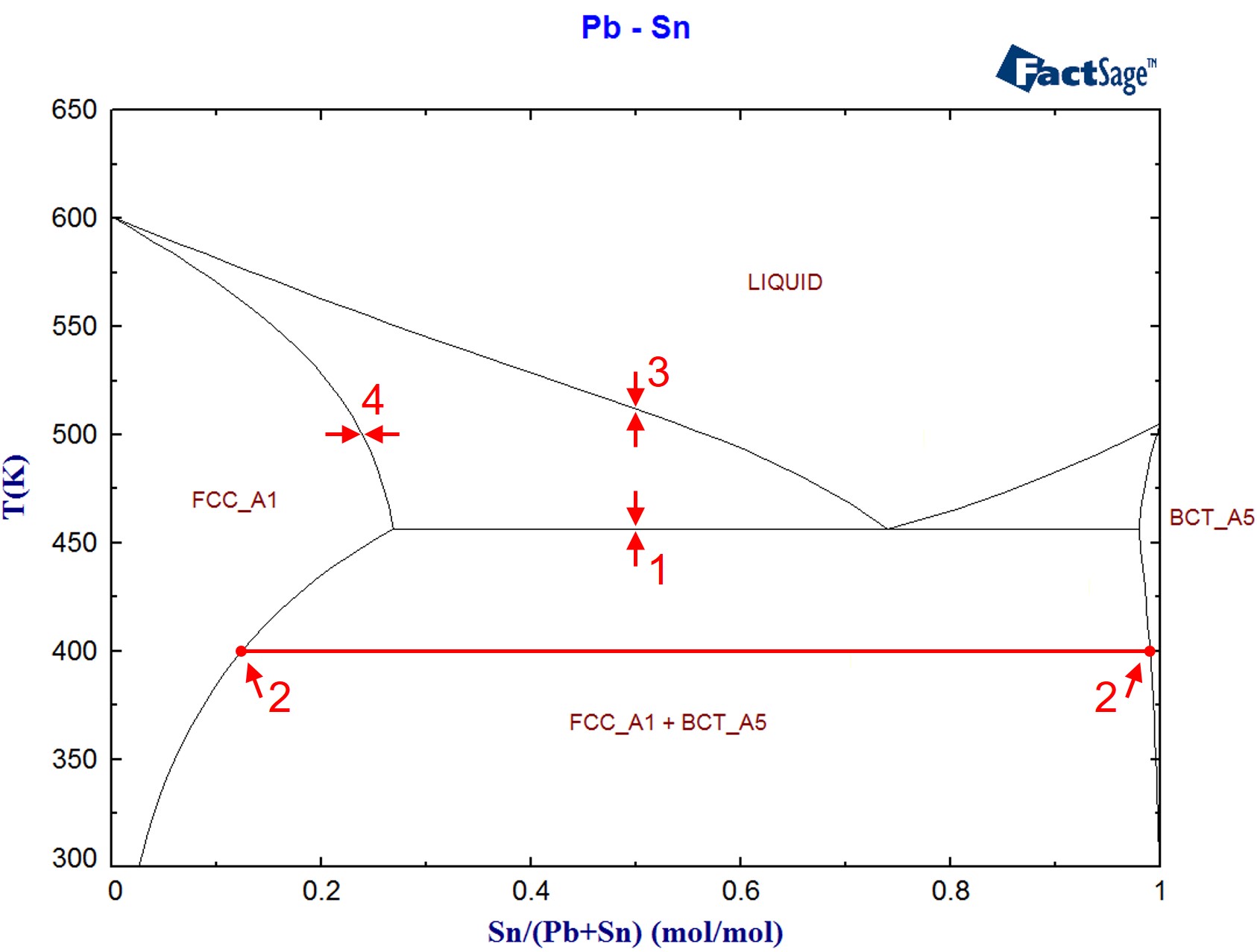
A phase 0 stage, so named because it is positioned between preclinical and phase I stages (Table 1), has the potential to improve preclinical candidate selection by applying approaches such as subtherapeutic microdosing (100 µg or less) to provide human in vivo data on pharmacokinetics, pharmacodynamics and target engagement earlier in the development process than with traditional approaches. A key strategy to reducing attrition rates is to improve the quality of candidates entering clinical development 6, 7, 8. Reducing attrition in clinical development is the single most important challenge in increasing pharmaceutical industry productivity 1, 2, 3, 4, 5.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
